Huidcarcinoom, vooral bij orgaantransplantatiepatiënten
De patiënten
Het risico op plaveiselcelcarcinoom is dramatisch verhoogd bij patiënten met een aangetast immuunsysteem, zoals ontvangers van orgaantransplantaties. Mensen met een niertransplantatie die huidlaesies ontwikkelen vormen een speciale focusgroep in onze kliniek: zij worden nauwlettend gevolgd om mogelijke huidcarcinomen in een vroeg stadium op te sporen voor een adequate en zo weinig mogelijk invasieve behandeling. De dominante risicofactor hier is blijkbaar de levenslange immunosuppressieve behandeling om afstoting van het transplantaat te voorkomen. Omgevingsfactoren, zoals blootstelling aan zonlicht en HPV-infectie, en genetische factoren, zoals een blanke huid en polymorfismen in cruciale genen, zijn echter ook van groot belang voor het risico op carcinoom bij ontvangers van orgaantransplantaten.
…De patiënten
Het risico op plaveiselcelcarcinoom is dramatisch verhoogd bij patiënten met een aangetast immuunsysteem, zoals ontvangers van orgaantransplantaties. Mensen met een niertransplantatie die huidlaesies ontwikkelen vormen een speciale focusgroep in onze kliniek: zij worden nauwlettend gevolgd om mogelijke huidcarcinomen in een vroeg stadium op te sporen voor een adequate en zo weinig mogelijk invasieve behandeling. De dominante risicofactor hier is blijkbaar de levenslange immunosuppressieve behandeling om afstoting van het transplantaat te voorkomen. Omgevingsfactoren, zoals blootstelling aan zonlicht en HPV-infectie, en genetische factoren, zoals een blanke huid en polymorfismen in cruciale genen, zijn echter ook van groot belang voor het risico op carcinoom bij ontvangers van orgaantransplantaten.
Onderzoek
Onze afdeling onderzoekt de rol van genetische en omgevingsfactoren, waaronder medicatie, bij het ontstaan van huidcarcinomen, vooral in relatie tot het verhoogde risico bij niertransplantatiepatiënten. Bij deze onderzoeken werken wij nauw samen met de afdelingen Nefrologie en Immuno-Hematologie, Medische microbiologie, Pathologie, Klinische Epidemiologie, Medische Genetica, Toxico-Genetica en Medische Statistiek. Het beloop van huidcarcinoom wordt bestudeerd door een genoombrede analyse van genexpressie in verschillende stadia van tumorontwikkeling (vergelijkbaar met studies in de onderzoekslijn ‘Lymfoom’), de groei van tumoren in gekweekte 3D-huidmodellen (zie onderzoekslijn ‘Menselijke Huidmodellen”) en in muismodellen. In vitro organotypische modellen van menselijke actinische keratose en plaveiselcelcarcinoom kunnen uitstekende experimentele vehikels blijken te zijn bij het ontwikkelen van goed gerichte antitumormedicatie zonder enige beperking en met directe menselijke relevantie, waardoor de huidige diermodellen worden vervangen.
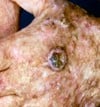
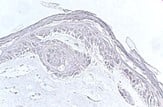
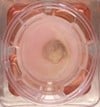
Figuur 1: links weergegeven) Menselijk huidmodel gegenereerd met een SCC-biopsie; Midden) Dwarsdoorsnede van het huidmodel na 4 weken kweken (invasief fenotype is aanwezig) en rechts) Hand van een patiënt met een SCC.
Doel
Het uiteindelijke doel van deze onderzoekslijn is het identificeren van de tumorspecifieke veranderingen in de relevante huid(stam)cellen en in signaaltransductieroutes die kenmerkend en essentieel zijn voor de ontwikkeling van plaveiselcelcarcinoom (SCC) en zijn voorloper, actinische keratose (AK). Deze informatie kan ons in staat stellen cruciale moleculaire doelwitten te identificeren voor klinische interventies die specifiek de tumor of de vroege ontwikkeling ervan beïnvloeden, zonder bijkomende schade aan gezond weefsel.
Studies bij patiënten en met menselijke cellen
Bij ontvangers van een niertransplantaat (RTRs) bestuderen we risicofactoren voor de ontwikkeling van SCCs om patiënten te identificeren die een verhoogd risico lopen, en om mogelijke pathogene mechanismen te identificeren die bijdragen aan de ontwikkeling van SCC. De afgelopen jaren hebben we ons geconcentreerd op de rol van cutane HPV-infecties, maar in vivo studies bleken te worden belemmerd door de schaarsheid van HPV in de niet-neoplastische huid in combinatie met een gebrek aan technieken om de geïnfecteerde huidcellen te identificeren.
In vitro-onderzoeken met menselijke huidcellen die zijn getransfecteerd met HPV-genconstructen hebben sterk uiteenlopende effecten van verschillende soorten HPV aan het licht gebracht (bijvoorbeeld verhoogde overleving of onderdrukking van apoptotische reacties bij blootstelling aan UV), wat ondersteuning biedt aan het concept van ‘hoog risico’ versus ‘laag risico’ types.
SCC's, AK's en normale huid van RTR's zijn geanalyseerd op genuitdrukkingen en chromosomale afwijkingen. Een groot aantal genen bleken op verschillende manieren tot expressie te komen tussen deze drie groepen monsters en deze verschillen waren moeilijk ondubbelzinnig in verband te brengen met enige cruciale of dominante verandering in de signaalroute. Een activatie van het RAS-traject van AK naar SCC bleek het meest opvallende resultaat. Chromosomale afwijkingen in SCC uit onze RTR's lijken minder te zijn dan eerder gerapporteerd voor SCC uit immunocompetente mensen. Dit kan een (gedeeltelijk) andere pathogenese van SCC bij RTR's weerspiegelen.
Meer recentelijk is onze aandacht gevestigd op immuunreacties die gericht zijn op het transplantaat, variërend van afstoting tot tolerantie, en de kracht van de immuunuitdaging, bijvoorbeeld verhoogd door gelijktijdige transplantatie van nieren en pancreas in vergelijking met transplantatie van alleen een nier. De overeenkomstige variaties in immuunreacties lijken het risico op PCC te beïnvloeden.
Studies bij muizen
Bij muizen bestuderen we vroege oncogene veranderingen veroorzaakt door UV-straling en de rol van stamcellen bij de vorming en instandhouding van tumoren. De effecten van verschillende immunosuppressieve behandelingen (met azathioprine, ciclosporine, tacrolimus, mycofenolaatmofetil en rapamycine) op UV-carcinogenese worden beoordeeld in een muismodel en vergeleken met observaties over het carcinoomrisico in RTR's), en met kortetermijneffecten in menselijke huidmodellen ( “equivalenten van de menselijke huid”, HSE’s). Verrassend genoeg en in tegenstelling tot het verhoogde risico op huidcarcinomen bij RTR's, hebben we geen verplichte versnelling van UV-carcinogenese gevonden door behandeling met de verschillende immunosuppressiva. In tegenstelling tot eerdere experimenten vonden we een gebrek aan overeenstemming tussen het effect van een immunosuppressivum op het aantal vroege klonen met tot overexpressie gebrachte mutant-p53 en de daaropvolgende tumorontwikkeling (dat wil zeggen dat een toename van het aantal klonen zich niet noodzakelijkerwijs vertaalde in een verbeterde tumorontwikkeling en vv-afnames in klonen vertaalde zich niet in een vertraagd begin van de tumor). Na het ontbreken van een verbeterde UV-carcinogenese door immunosuppressiva, willen we het effect van een volledige orgaantransplantatie op UV-carcinogenese onderzoeken.

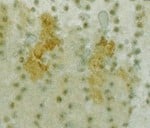
Figuur 2: Getoond worden mutant-p53-positieve cellen aanwezig in de linker) interfoliculaire foci van de bestraalde muizenhuid en rechts) SCC.
In vervolg op onze eerdere studie naar de rol van rustende stamcellen in de ontwikkeling van carcinomen als gevolg van chronische lage UV-blootstelling van de huid, zullen we de rol onderzoeken van nieuw prolifererende stamcellen in UV-carcinogenese; Dit doen wij in samenwerking met prof. Hans Clevers (Hubrecht Lab, Ultrecht, Nederland) die – samen met de groep van prof. Tofgard (Karolinska Inst., Stockholm, Zweden) – deze (Lgr-5 en -6 positieve) stamcellen ontdekte in de haarzakjes van de huid .
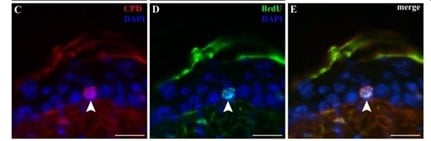
Figuur 3: Er worden dwarsdoorsneden weergegeven van een epidermis met CPD-accumulatie in BrdU-vasthoudende cellen/stamcellen (pijl).
UV en vitamine D
In samenwerking met de afdeling Menselijke Genetica onderzoeken we het effect van UV-blootstelling van de huid, en het effect van vitamine D3 op darmkanker in een transgeen APC-mutant muismodel. Verdere studies naar de fotosynthese van vitamine D3 in de huid zijn belangrijk omdat de netto productie onder verschillende omstandigheden (zonnespectra) niet voldoende bekend is (de wisselwerking tussen de 6 betrokken fotochemische reacties moet in detail worden bestudeerd, vooral voor golflengten boven 310 nm) . Deze informatie is nodig om mensen beter te informeren over adequate blootstelling aan vitamine D-synthese met een minimale toename van het risico op huidcarcinomen. De actieve metaboliet van vitamine D3 (1,25 dihydroxyvitmain D3) bindt ook aan de vitamine D-receptor in huidcellen en oefent waarschijnlijk een remmend effect uit op de groei van huidcarcinomen.
Hoofdonderzoekers
- FR de Gruil
- JN Bouwes Bavinck
Huidcarcinomen zijn de meest voorkomende kwaadaardige aandoeningen bij blanke bevolkingsgroepen en blootstelling aan ultraviolette straling van de zon lijkt een belangrijke etiologische factor te zijn. UV-straling beschadigt DNA, wat mutaties in genen veroorzaakt die huidcellen kwaadaardig kunnen maken. Variaties in genetische achtergrond, nl. bij pigmentatie en herstel van beschadigd DNA, de gevoeligheid voor de vorming van huidcarcinomen bepalen. Bepaalde typen humaan papillomavirus (HPV) kunnen de gevoeligheid verhogen. Naast deze directe kankerverwekkende gevolgen beïnvloedt UV-straling het immuunsysteem, waarbij met name de cellulaire immuniteit tegen huidcarcinomen kan worden onderdrukt. Dergelijke immuunmodulerende effecten worden in de dermatologische kliniek gebruikt bij de fototherapie van huidziekten, met name psoriasis, maar deze immunologische effecten kunnen ook “zonneallergie” van de huid veroorzaken (“polymorfe lichtuitbarsting”, PLE). Van algemeen voordeel voor onze gezondheid is de UV-gestuurde synthese van vitamine D3 in de huid, wat onder andere kan leiden tot remming van de tumorgroei.