Genome Engineering
The most promising genes resulting from these screens are being studied in further detail using genetic and molecular approaches in worms, fish and mammalian cells.
Our primary research interest is how organisms, cells or genomes maintain genetic integrity, and over the last few years we have used the model organism C. elegans to identify and characterize genes whose loss of function results in an enhanced level of spontaneous mutagenesis, so-called “mutator” genes. In parallel to cloning these genes via conventional genetic approaches, we have developed sensitive in vivo assays to detect specific types of genomic instability, such as micro-satellite instability, G4 DNA instability and DNA transposition. In addition, we developed reporter-based single worm assays that allow us to monitor error prone repair of DNA double strand breaks. The rationale has been to create tools that could optimally benefit from the strength of the C. elegans system, a fast genetic animal system that, since the discovery of RNAi, has become amenable to systematic genome-wide reverse genetic approaches. By combining these novel assays with genome-wide RNAi, we aim to identify the complement of genes that are required to maintain genomic integrity.
We have also progressively worked towards more efficient and faster RNAi screening methods with the aim to perform synthetic lethal screens in an animal system. Two genes are called synthetic lethal when cell death is manifested if both genes are inactivated, whereas inactivation of only one gene does not lead to loss of viability. Such interactions can have great clinical potential in cases where genes are causally linked to tumorigenesis. To obtain proof of principle for our experimental setup, we have used synthetic lethal screening to identify genes that protect the genome from DNA double strand breaks generated by transposon excision. The protocol to perform genome-wide RNAi screens in a 96-well liquid platform has also been used to directly identify modifiers of DNA double strand break responses by scoring for ionizing radiation sensitivity.
…We have also progressively worked towards more efficient and faster RNAi screening methods with the aim to perform synthetic lethal screens in an animal system. Two genes are called synthetic lethal when cell death is manifested if both genes are inactivated, whereas inactivation of only one gene does not lead to loss of viability. Such interactions can have great clinical potential in cases where genes are causally linked to tumorigenesis. To obtain proof of principle for our experimental setup, we have used synthetic lethal screening to identify genes that protect the genome from DNA double strand breaks generated by transposon excision. The protocol to perform genome-wide RNAi screens in a 96-well liquid platform has also been used to directly identify modifiers of DNA double strand break responses by scoring for ionizing radiation sensitivity.
The most promising genes resulting from these functional genomic screens are being studied in further detail using genetic and molecular approaches in worms, fish and mammalian cells.
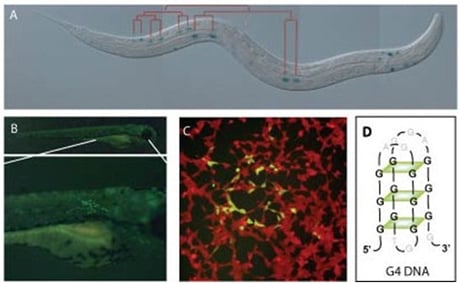 Figure 1. DNA instability visualized in the model systems we employ: the nematode C. elegans (A), the fish D. rerio (B) and human cells (C). Alternatively folded DNA, such as G4 DNA (D) can be a cause of DNA instability leading to gross chromosomal rearrangements. Underlying mechanisms of protection are subject of study.
Figure 1. DNA instability visualized in the model systems we employ: the nematode C. elegans (A), the fish D. rerio (B) and human cells (C). Alternatively folded DNA, such as G4 DNA (D) can be a cause of DNA instability leading to gross chromosomal rearrangements. Underlying mechanisms of protection are subject of study.
Key publications
Our Team
Prof.dr. Marcel Tijsterman
Principal Investigator / Professor Genome stability
Lejon E.M. Kralemann
Researcher (IBL)